
Eighth group: This group is full of anomalies. Dissimilar elements placed in the same groups: The elements in the subgroup A and B of a particular group show different physical and chemical properties but still they are placed in the same group in Mendeleev’s periodic table. Similar elements are not placed in the same groups: The elements like silver and thallium, barium and lead, copper and mercury showing similar properties are placed in a different group in Mendeleev’s periodic table.ħ. That is, potassium should be placed in the zero group while argon in the IA group.Ħ. If the criterion of atomic weight is strictly followed, the potassium should be placed before the argon. Similarly, tellurium (atomic mass \(= 127.61\)) is placed before Iodine (atomic mass \(= 126.91\)). Anomalous pair of elements: Several elements are not arranged in the increasing atomic mass, i.e., not placed according to Mendeleev’s periodic law.įor example, argon (atomic number \( = 39.9\)) is placed before potassium (atomic number \(= 39.1\)). But this was not mentioned in Mendeleev’s periodic table.ĥ. Elements with variable valency: A large number of elements show more than one valency. 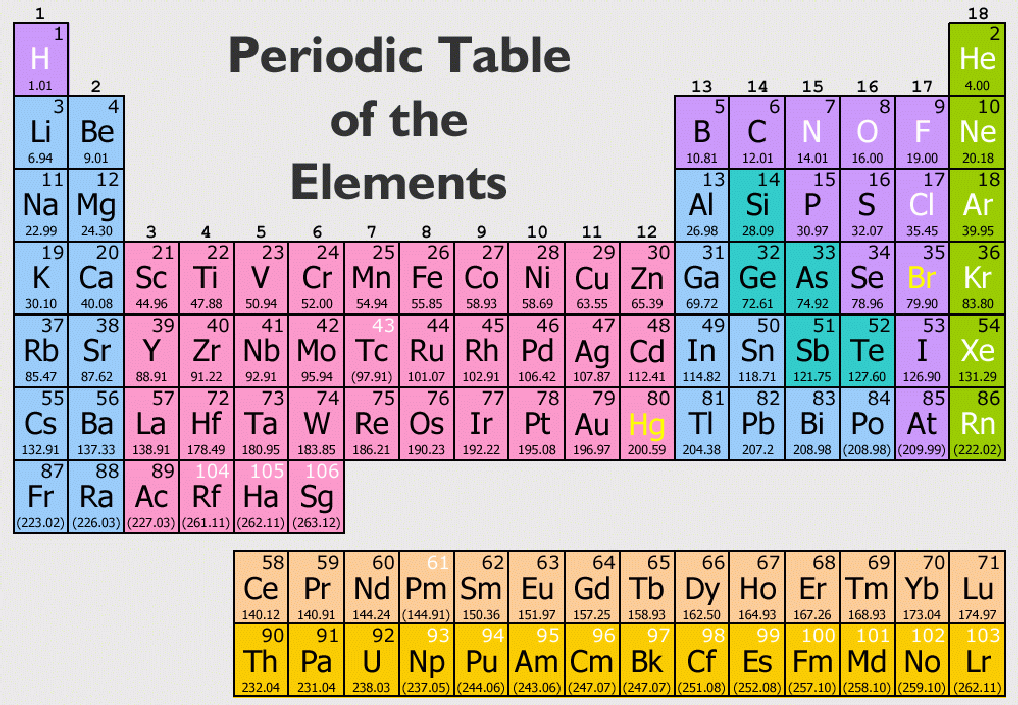
These arrangements are anomalous in Mendeleev’s periodic table since they are not following Mendeleev’s periodic law.Ĥ. Position of lanthanides and actinides: The \(14\) elements following lanthanum atomic number \(58\) to \(71\) are lanthanides and \(14\) elements following actinium, atomic number \(90-103\) are actinides. However, these have not been given in different places in the periodic table.ģ. But this will be inconsistent because isotopes possess similar chemical properties.Įxample: Protium, deuterium, and tritium with atomic weights \(1,\) \(2,\) and \(3\) respectively should be placed at different places. Therefore, isotopes of an element should be placed at different places. Position of isotopes: In Mendeleev’s periodic table, elements are arranged in the increasing atomic masses. But, hydrogen has been placed in the IA group in this periodic table.Ģ. Since hydrogen exhibits both the properties of alkali metals (IA group) and halogens (VIIA group). Position of hydrogen: Based on properties, hydrogen should have been placed in both group IA and group VIIA. The first period with only two elements is called the shortest period.The first half of the elements are placed in the upper left corner, and the second occupies the lower right corner in each box. To accommodate more elements, periods \(4, 5, 6,\) and \(7\) are divided into two halves. There are seven periods (numbered from \(1\) to \(7\)). 
The zero group contains elements belonging to inert (noble) gases, and the elements present have zero valencies. Group VIII consists of \(9\) elements which are arranged in triads. The elements that are placed on the left-hand side of each group constitute subgroup A, while those on the right-hand side form subgroup B. The elements belonging to the first \(7\) groups have been divided into subgroups designated as A and B based on similarity in properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
